Nitroso
Nitrosamines are a group of chemical compounds that contain the nitroso functional group (NO) attached to an amine group (-NH). They can form in various ways, including through industrial processes, combustion, and certain chemical reactions, particularly involving nitrites and secondary amines. Nitrosamines are of concern because Many nitrosamines (N-Nitrosamines) are potent genotoxins; some have been classified as possible human carcinogens, although the degree of risk can vary depending on factors such as the amount of exposure and individual susceptibility. Synchemia Research Chemical is dedicated to thoroughly investigating the formation of nitroso impurities in APIs, including those related to the manufacturing processes. This approach indicates a proactive stance toward quality control and regulatory compliance. By exploring all potential sources of nitroso impurities, including those arising from the production processes themselves, Synchemia Research Chemical demonstrates a commitment to ensuring the purity, safety, and efficacy of their pharmaceutical products. Nitrosoamines, including N-Des-methyl Nitroso impurities, are important in pharmaceutical research and development due to their potential impact on drug safety and efficacy. Understanding and controlling the presence of these impurities is crucial for ensuring the quality and safety of pharmaceutical products. Can N-Nitrosamines exist in two different stable forms? N-nitrosamines substituted with two different alkyl groups can exist in two different stable forms distinguishable by several analytical methods. The two rotamers may differ in their overall dipole moment and therefore may posses different chemical and physical properties. Why do I see two peaks when analyzing N-Nitrosamines by HPLC or GC? HPLC and GC)analysis of N-nitrosamines may show two rotamers of the N-nitrosamine as two separate peaks. Whether two peaks are observed depends on the structure of the N-nitrosamine (i.e., symmetric vs. asymmetric) as well as the separation power of the analytical method.
We are the Synchemia Research Chemical specializes in manufacturing and exporting various nitrosoamine compounds, particularly focusing on N-Des-methyl Nitroso impurities of various APIs and nitroso compounds of direct APIs that contain secondary amines. By specializing in the synthesis and export of these compounds, Synchemia Research likely plays a significant role in supporting the pharmaceutical industry's efforts to meet regulatory requirements and ensure the highest standards of quality and safety in drug manufacturing.
 Download catalogue
Download catalogue
Pharmaceutical Reference Standards
N-Nitroso Sumatriptan EP Impurity A
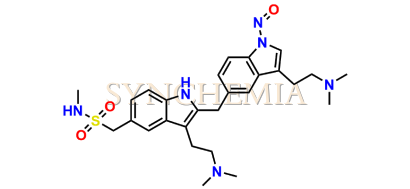
| CAT No | SRC-N014836 |
| CAS No | NA |
| Mol.F. | C27H36N6O3S |
| Mol.Wt. | 524.7 |
| INV STATUS | CUSTOM SYNTHESIS |
N-Nitroso Itraconazole Impurity 1
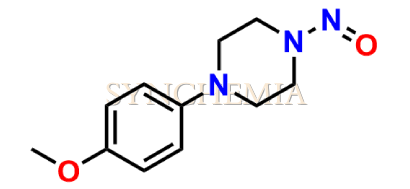
| CAT No | SRC-N014837 |
| CAS No | 75051-59-1 |
| Mol.F. | C11H15N3O2 |
| Mol.Wt. | 221.3 |
| INV STATUS | IN STOCK |
N-Nitroso Sumatriptan EP Impurity D
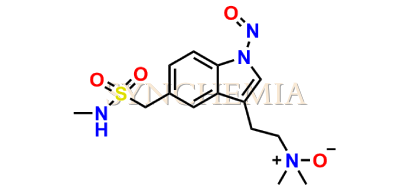
| CAT No | SRC-N014838 |
| CAS No | NA |
| Mol.F. | C14H20N4O4S |
| Mol.Wt. | 340.4 |
| INV STATUS | CUSTOM SYNTHESIS |
N-Nitroso Sumatriptan EP Impurity E
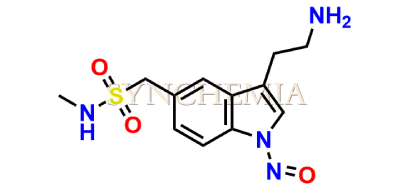
| CAT No | SRC-N014839 |
| CAS No | NA |
| Mol.F. | C12H16N4O3S |
| Mol.Wt. | 296.3 |
| INV STATUS | CUSTOM SYNTHESIS |
N-Nitroso Rivaroxaban Dioxazolidine Impurity
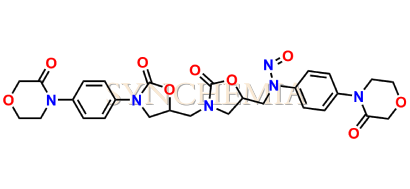
| CAT No | SRC-N014840 |
| CAS No | NA |
| Mol.F. | C28H30N6O9 |
| Mol.Wt. | 594.6 |
| INV STATUS | CUSTOM SYNTHESIS |
Rivaroxaban Amino Acid Phthalimide Nitroso Impurity
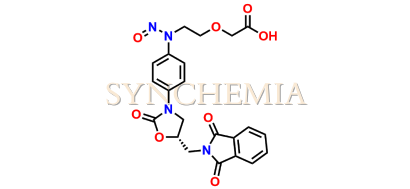
| CAT No | SRC-N014841 |
| CAS No | NA |
| Mol.F. | C22H20N4O8 |
| Mol.Wt. | 468.4 |
| INV STATUS | CUSTOM SYNTHESIS |
Rivaroxaban Nitroso Impurity 13
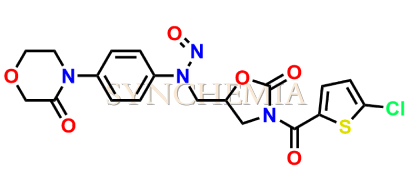
| CAT No | SRC-N014842 |
| CAS No | NA |
| Mol.F. | C19H17ClN4O6S |
| Mol.Wt. | 464.9 |
| INV STATUS | CUSTOM SYNTHESIS |
Rivaroxaban Nitroso Impurity 14
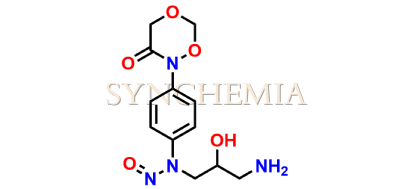
| CAT No | SRC-N014843 |
| CAS No | NA |
| Mol.F. | C12H16N4O5 |
| Mol.Wt. | 296.3 |
| INV STATUS | CUSTOM SYNTHESIS |
Rivaroxaban Nitroso Impurity 15
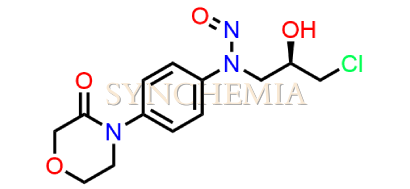
| CAT No | SRC-N014844 |
| CAS No | NA |
| Mol.F. | C13H16ClN3O4 |
| Mol.Wt. | 313.7 |
| INV STATUS | CUSTOM SYNTHESIS |
Rivaroxaban Nitroso Impurity 16
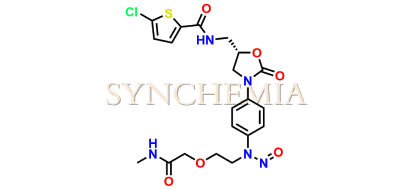
| CAT No | SRC-N014845 |
| CAS No | NA |
| Mol.F. | C20H22ClN5O6S |
| Mol.Wt. | 495.9 |
| INV STATUS | CUSTOM SYNTHESIS |
Let’s advance your research together.
Partner with us for specialized chemical materials and synthesis services that enable pharmaceutical innovation.
 info@synchemia.com | sales@synchemia.com
info@synchemia.com | sales@synchemia.com  9766925136 | 72
9766925136 | 72 



